(Note: this is part of a series of posts, in which I attempt explain a basic physics concept and put it in a wider context. The first Physics Quantum introduced the basic physics of ideal pendulums undergoing small oscillations; the second extended the idea to large oscillations and added in air resistance. As I promised, this is the third pendulum-related post, connecting the physics of oscillators to quantum mechanics, where things don’t look like masses on the ends of strings anymore.)
Back in May, psychologist-magician Richard Wiseman (ain’t that a great combo?) posted a great video that has since gotten a lot of play around the internet :
Here’s what I had to say about it at the time. To summarize, I pointed out that the patterns we see are emergent, based on the relative lengths of the pendulum strings. They aren’t interacting with each other, but the fact that the lengths of the strings are mathematically related to each other creates the beautiful waveforms. In other words, despite there being no direct interaction, there is a kind of indirect relationship that is intriguing. A similar insight led Max Planck and Albert Einstein to the beginnings of quantum mechanics; in this post, I’ll start from a modern understanding of quantum oscillators and work backwards to show how these pendulum waves connect to all sorts of interesting phenomena.
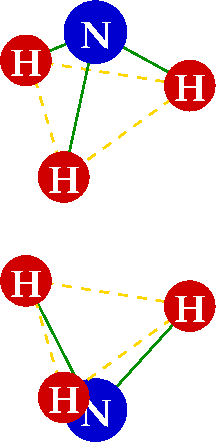
An ammonia molecule is a simple quantum mechanical oscillator that looks kind of like a pendulum if you use a bit of imagination. As the figure shows, ammonia consists of three hydrogen atoms arranged in an equilateral triangle, with a nitrogen atom making a total pyramid shape. This molecule appears to be somewhat asymmetric, though! There’s no real reason to draw the nitrogen “on top” of the pyramid rather than “below” the hydrogen atoms — it’s like having the pendulum bob on the “left side” vs. the “right side”. Those two positions of the nitrogen atom or pendulum bob are energetically equivalent: they represent the same physical state.
Enter Emmy Noether and her theorem: if there is a symmetry in energy between two states, there must be a symmetry in time too. That’s the fancy way to put it; the simple version is that there has to be oscillation! Just as a pendulum swings back and forth, the nitrogen atom must move back and forth from the “above” to “below” the triangle of hydrogen atoms. In fact, it is impossible for the nitrogen atom to stay in one place without an outside influence, even though there is an energy barrier at the location of the hydrogen triangle — the nitrogen atom must tunnel through to complete its oscillation. (Tunneling is of course an interesting phenomenon in its own right, which I will leave for another day. Here’s a fun 1-minute explanation.) A major difference between the ammonia molecule and a pendulum is that the pendulum swings because of gravity; the ammonia molecule will oscillate entirely on its own.

Of course you can force the nitrogen atom to prefer one position over the other, in the same way you can put an external force onto a pendulum. If you put electrical pulses through a bunch of ammonia gas, you can stimulate transitions, forcing the molecules to emit photons of a particular frequency in the microwave part of the spectrum. This system is called a maser: microwave amplification by the stimulated emission of radiation — the microwave version of a laser. Charles Townes, J. P. Gordon, and H. J. Zeiger built the first ammonia maser in 1953; masers from other molecules have been found in interstellar gas clouds.
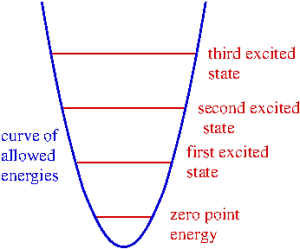
In general, quantum oscillators can only have certain specific internal energy values. After all is what makes them quantum: they can only absorb and emit certain quantities of energy. As we saw with the ammonia molecule, even when left alone a quantum oscillator will fluctuate at a specific energy, which goes under the fancy name of zero point energy. (In the Pixar film The Incredibles, the villain Syndrome uses zero point energy to fling the heroes around effortlessly, which is obviously a bit of license on the screenwriters’ part.) Quantum oscillators come in many forms, from simple molecules to crystalline solids. The Einstein model of a solid is based on a big lattice of independent quantum oscillators… and here’s where we get back to that wonderful pendulum video at the beginning of the post.
Each oscillator in the solid is identical, in the sense that each one has the same set of energy levels and zero point energy. This means that when any oscillator transitions from one energy level to another, it will emit or absorb the same amount of energy as any other in the solid. Even though the oscillators don’t interact with each other directly, because they have the same energy levels they will act in concert under the right conditions, just as the pendulums’ lengths in the video are related to each other, so they create patterns.
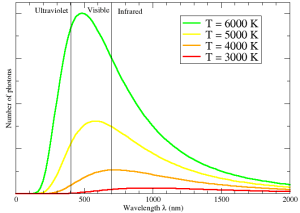
The pattern created by oscillators in the Einstein model is one of light, since the energy absorbed and emitted is in the form of photons, and the details depend on the temperature. Temperature is a measure of average kinetic energy — the energy of motion — so the higher the temperature, the more energy is available to put into the oscillators. As a result, an interesting pattern of light emerges, known as the thermal spectrum. As I wrote in an earlier post about why there are no green stars, hotter objects emit more light than cooler objects, again because there is more energy available to the oscillators that create photons.
We’ve come a long way from a weight on the end of a string, but so many of the basic concepts are the same. Oscillators are some of the most important objects in all of physics, tying together ideas you learned in elementary school to cutting-edge research in theoretical physics, biology, networks, and so on. Galileo’s pendulum may have been the starting point, but we are hardly finished with understanding oscillators. Expect more in weeks to come!

14 responses to “From Pendulums to Quantum Oscillators”
[…] Galileo's Pendulum The Pendulum is Mightier Than the Sword « From Pendulums to Quantum Oscillators […]
[…] Every student in introductory astronomy learns the mnemonic “Oh Be a Fine Girl/Guy, Kiss Me”, which is a way to remember the spectral classification of stars. Here’s how it works: the color of a star is determined by its temperature, as I outlined previously. […]
[…] discussed how unconnected oscillators can appear to link up, creating patterns and giving rise to phenomena such as lasers and the spectra of stars. We’ve come a long way from masses on strings: we can understand oscillators as any system […]
[…] both in mass and size; as a result, their light is much less intense. Stars emit light according to a thermal spectrum: the hotter a star, the more white it appears and the more light it emits, while a cooler star of a […]
[…] of Virginia. I will write a post about Foucault pendulums soon(ish), as a continuation of the general oscillator […]
[…] masses from each other and in their natural state are a combination; in this sense they are similar to the ammonia molecule I wrote about in an earlier post. To summarize, in the case of ammonia, there are two states that are equally probable (the nitrogen […]
[…] iron becomes magnetized, that symmetry is lost: one direction is more important than the others. An ammonia molecule is symmetrical, and that’s the source of its oscillations, but not all molecules work that way. Organic molecules (in standard scientific usage) are […]
[…] temperature of the host star: the hotter the star, the more it emits light of all wavelengths. This includes infrared light, which is the most important for keeping a planet warm. For a cooler, […]
[…] August: “Centrifugal Forces and Trojan Horses” and “From Pendulums to Quantum Oscillators“ […]
[…] I mentioned in earlier posts on the subject, the color of a star is dictated by its surface temperature. Hot stars appear blue or blue-white, while cool stars appear red. However, no matter how hot or […]
[…] there is a star inside or not. Without further work, we don’t know how big the star is or how hot it is or even where it lies inside the box, but we know how much total light it […]
[…] as they vary in time, so it exhibits harmonic motion, like a pendulum swinging back and forth. Like the ammonia molecule I described in an earlier post, however, the oscillations obey the rules of quantum physics, so they themselves […]
[…] but those aren’t common in astronomy.) Each type of atom or ion has its own unique spectrum, as described by quantum mechanics, so astronomers can identify the chemical composition of gas clouds in distant galaxies—even if […]
[…] object. Humans are approximately blackbodies: our metabolic processes make us glow in the infrared. Stars are much closer to being ideal blackbodies, but the Universe as a whole is nearly perfect. If photons have mass, then that mass would slightly […]